4 reasons to stop using spreadsheets for trial data collection (Infographic)

Organizations, which still use spreadsheets for trial data collection, seem to increase the complexity behind the studies they conduct. Sadly, the intricacy of the advanced softwares created for data collection, makes many organizations still use spreadsheets. Nonetheless, there are various solutions emerging on the market, which can help resolve this problem.
Clinical trial management systems (CTMS) are those innovative software solutions, which help storing the entire data in one place. Moreover, unlike Microsoft Excel’s spreadsheets, they have been specifically designed to collect such information.
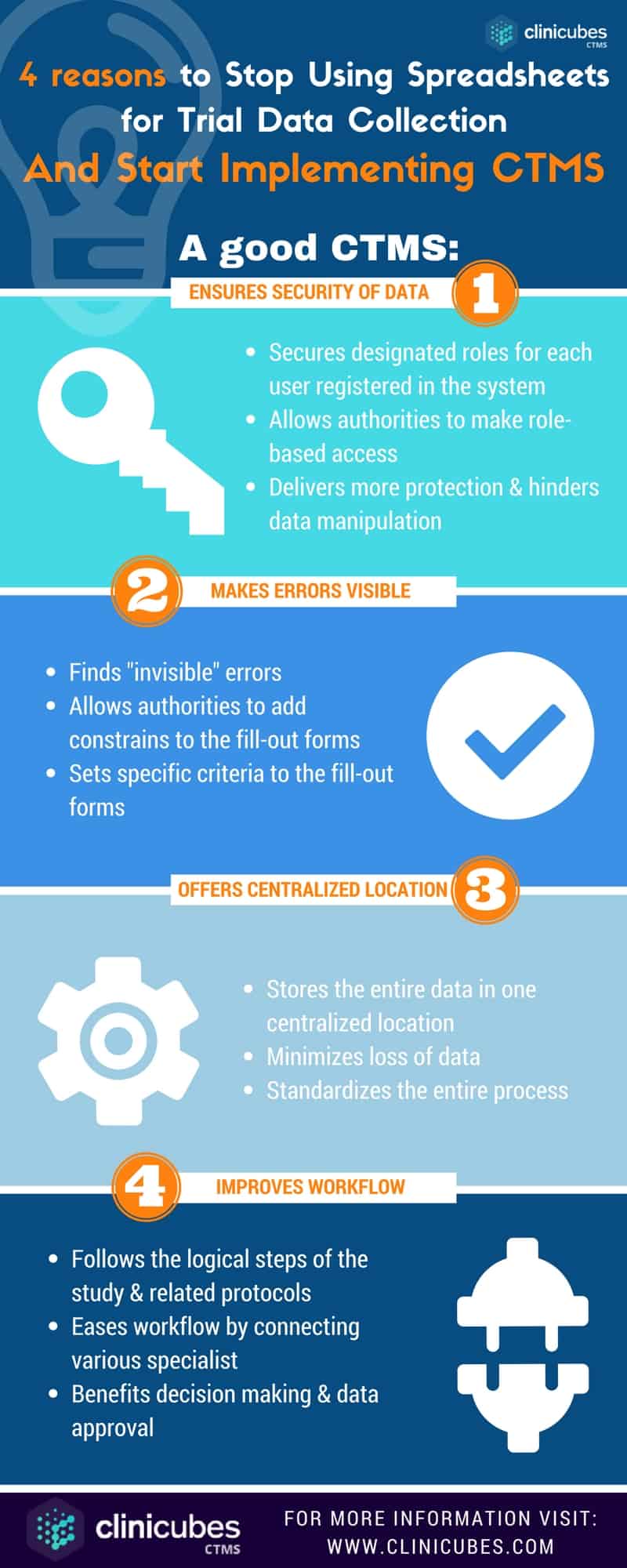
The here presented infographic shows how a good CTMS solves four commonly faced problems that no spreadsheet can:
1. The insecurity of data
2. The “invisibility” of errors
3. The lack of centralized location to store the data
4. The inefficacy of the workflows
Therefore, the clinical trial management system:
1. Ensures security of data
In contrast to spreadsheets, which are usually accessible from anyone, CTMS not only allows multiple users, but also secures designated roles for each one registered in the system. The software stores the entire information in one centralized location and authorities make the role-based access to it. This is how a CTMS delivers more protection and hinders data manipulation.
2. Makes errors visible
Errors in trial data collection usually go unnoticed when utilizing spreadsheets. The innovative software solutions allow authorities to add constrains to the forms, which other specialists will fill out. When the criteria are set, there is no place for mistakes. This is one of the most beneficial features of an adaptive CTMS.
3. Stores the entire data in one centralized location
The entire information related with the trial is entered into the software. This allows for minimization of the cases when data is lost, misplaced or cut out. On the contrary, spreadsheets are usually stored in different folders, making them unfeasible and hard to locate. Secondly, there might be more than just one copy. Clinical trial management systems are advantageous in a way that they standardize the entire process, identify the lastly updated document and store everything in one location.
4. Improves workflow
As a specialized software, the CTMS is designed to manage clinical trials from start to end. It follows the logical steps of the study and related protocols. It eases workflow by connecting various specialist: from sponsors, CROs, data managers, to everyone involved in the research process. This benefits decision making, data approval and upload of new versions of the documents, which are instantly available to anyone. The insufficiency of spreadsheets is that they weren’t designed initially to manage clinical data. Thus, they demolish the natural workflow. The success of a clinical trial depends on the functionality and workflow of all processes involved.
Using innovative software solutions like CTMS may be challenging. This is why it must be adaptable and user-friendly. It must collect, store and have the option of verifying trial data. These systems have been specifically designed for this purpose, and it is undoubtedly number one reason why organizations use them instead of spreadsheets. Why waste more time then?
As one of the latest clinical trial management system, Clinicubes is an example for such software solution. Being easy-to-use and adaptive to organizational needs, it has been designed to simplify complexity behind any research project. It solves the four mentioned problems in a way that no other clinical trial management system can.
Become our partner
We are currently looking for partners for the implementation and customization of Clinicubes. Get in touch and take advantage of what our clinical trial management system offers.




